formulation vessels
with ultrafiltration unit

BASIC INFO
our equipment
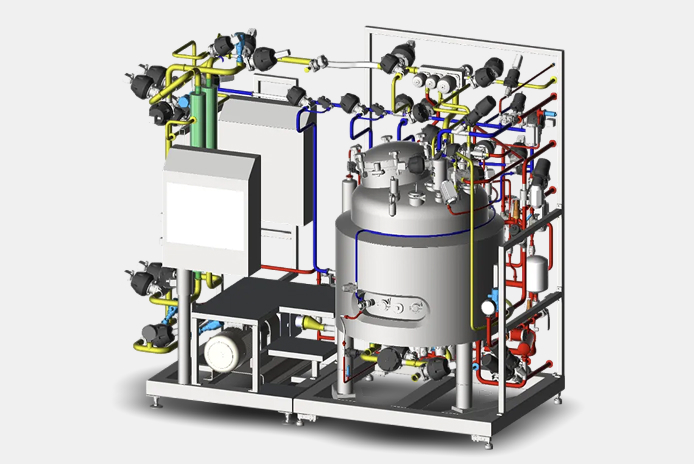
We offer delivery of formulation vessels according to specific customer requirements. We focus on stainless steel vessels with a volume of 50 to 2000 liters, usually equipped with a bottom magnetic stirrer.
The equipment can be delivered on a frame (skid unit) or assembled on site from separate elements.
Formulation vessels are equipped with a fully validatable control system according to GAMP 5 requirements with the possibility of connection to higher-level systems.
PROCESSES
The basic processes of the equipment include:
Sterilization of empty vessel, load and peripherals
Maintaining stable temperature and pressure in the vessel
Washing empty vessel and peripherals
Sampling including sterilization and washing of the sampling valve without impacting the content of the tank
Processes can be defined within the functional specification,
which is created during the design phase.
FUNCTION
ultrafiltration
We also offer formulation vessels with ultrafiltration through HFC (Hollow Fibre Cartridge). We currently offer a fully automated dual ultrafiltration system for maximum user comfort and safety. The system provides a simple way to preserve the HFC after the filtration process is complete.

HOW WE PROCEED
process of implementation
Unless the customer requests otherwise, we follow the steps of the regular "V" model and prepare all necessary documentation according to GMP requirements.
User Specification (URS)
Collaboration with the customer on detailed specifications. Thorough initial preparation helps eliminate deviations during the design and implementation of the device.
Risk Analysis (RA)
It serves to identify risks affecting the quality and safety of the equipment. It also contains risks mitigation and is essential for testing the equipment in future stages of implementation.
Functional Specification (FS)
It contains a design of the key processes of the equipment. It is an important basis for the design phase and later for programming and start up.
Design Qualification (DQ)
Based on the requirement, the design can be qualified, and the identified deficiencies are incorporated into the project documentation. This way, deficiencies are minimized during implementation.
Factory Acceptance Testing (FAT)
Once the device is completed, the basic functions are tested with the customer's participation. The device is then sent to the site.
Connecting the device
Our team is able to provide on-site preparation of product pipelines and equipment connections. This service is completely optional.
Site Acceptance Testing (SAT)
After installation at the customer's site, testing is performed, followed by handover of the equipment.
Qualifications
Through our validation department, we can also provide final qualification of equipment at IQ, OQ and PQ levels. It is up to the customer to decide whether to carry out these activities themselves or to contact our team.
Documentation
The device is supplied with complete documentation according to GMP standards, including e.g. diagrams, material certificates, calibration certificates (including loop calibration), etc. The complete list is always defined in URS.
EFFECTIVE COLLABORATION
designing
As part of the design activity, not only a 2D diagram (P&ID) is created, but also a 3D model, which allows more efficient work on the project and communication with the customer.


PRECISE LOCATION
augmented reality (AR)
Thanks to the 3D model, we can also use augmented reality, which allows us to display the device in space at 1:1 scale. It is also suitable for simulating relocation and defining a suitable relocation route.
FEATURES
dead legs elimination
Use of m-blocks – for maximum possible elimination of dead legs and better serviceability of peripherals. At the same time, it allows addition and circulation of the product simultaneously. The product is added to the container automatically based on continuous level measurement.



SAFETY
light signalization
Light signaling - the device is equipped with backlighting, which performs two basic functions. Safety - where the color changes (from green to red) with increasing temperature during sterilization and informs the user about hot surface. Process - when the flashing light signalizing the need for operator's intervention. The light signaling was successfully registered as an utility model at the Industrial Property Office.
REFERENCES
see our projects
references
We are a reliable partner
not only for the pharmaceutical industry.

get in touch
You have an individual requirement
or an idea and you don’t know how to proceed?
Always something extra in order to help you.
Impartial view and experience
from dozens of companies.
From idea to implementation.
We are here to help you.